Identification
CAS Number
76513-69-4
Name
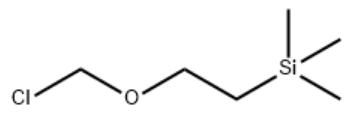
2-(Trimethylsilyl)ethoxymethyl chloride
Synonyms
(2-Chloromethoxyethyl)trimethylsilane
2-(Chlormethoxy)ethylsilan [German] [ACD/IUPAC Name]
2-(Chloromethoxy)ethylsilane [ACD/IUPAC Name]
2-(Chlorométhoxy)éthylsilane [French] [ACD/IUPAC Name]
[2-(chloromethoxy)ethyl]trimethylsilane
2-(Trimethylsilyl)ethoxymethyl chloride
278-483-4 [EINECS]
76513-69-4 [RN]
Chloromethyl 2-trimethylsilylethyl ether
MFCD00009919 [MDL number]
SEM-chloride
SEM-Cl
Silane, [2-(chloromethoxy)ethyl]trimethyl- [ACD/Index Name]
(2-(chloromethoxy)ethyl)trimethylsilane
[76513-69-4] [RN]
138786-65-9 [RN]
2- ethoxymethylchloride
2-(chloromethoxy)ethyl-trimethylsilane
2-(Chloromethoxy)ethyltrimethylsilane (stabilized with Diisopropylethylamine)
2-(Chloromethoxyethyl)trimethyl silane
2-(Chloromethoxyethyl)trimethyl silane|SEM-Cl
2-(Trimethsilyl)-ethoxymethyl chloride
2-(Trimethylsilyl)ethoxymethyl Chloride (SEM-Cl)
2-(Trimethylsilyl)ethoxymethylchloride
2-(trimethysilyl)-ethoxymethyl chloride
2-Chloroethoxy Methyl trimethyl silane
2-Chloromethyl 2-(trimethylsilyl)ethyl ether
95%
FS-3821
OS-7574
SEMCl
stabilized
SMILES
C[Si](C)(C)CCOCCl
StdInChI
InChI=1S/C6H15ClOSi/c1-9(2,3)5-4-8-6-7/h4-6H2,1-3H3
StdInChIKey
BPXKZEMBEZGUAH-UHFFFAOYSA-N
Molecular Formula
C6H15ClOSi
Molecular Weight
166.721
EINECS
278-483-4
Beilstein Registry Number
3587289
MDL Number
MFCD00009919
Properties
Appearance
Colourless to light yellow liquid
Safety Data
Symbol
Signal Word
Warning
Hazard statements
H226 – H314Precautionary Statements
P210 – P233 – P240 – P280 – P303 + P361 + P353 – P305 + P351 + P338WGK Germany
3
MSDS Download
Specifications and Other Information of Our
Purity
95% min
Water
≤2.0%
Single Impurity
≤3.0%
Total Impurities
≤5.0%
Shelf Life
2 years
Known Application
2-(Trimethylsilyl)ethoxymethyl chloride CAS 76513-69-4 (SEM-Cl) is an organosilicon compound commonly used as a protecting group in organic synthesis.
SEM-Cl is frequently used as a protecting group in organic synthesis, particularly for protecting hydroxyl (-OH) and amino (-NH2) groups. The use of protecting groups can prevent these functional groups from undergoing unwanted reactions during the synthesis process, thereby increasing the selectivity and efficiency of the synthesis.
In the synthesis of complex organic molecules, SEM-Cl can be used to temporarily protect certain functional groups. Once other parts of the synthesis are completed, the protecting group can be removed under specific conditions to restore the original functional group.
General View of Documents
Links
This product is developed by our R&D company Caming Pharmaceutical Limited (http://www.caming.com/).
Quick Inquiry
Fill out our inquiry form and one of our experts will be in touch with you shortly.