Identification
CAS Number
89-57-6
Name
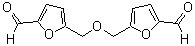
Mesalazine//Mesalamine//5-Aminosalicylic acid(5-ASA) CAS 89-57-6
Synonyms
201-919-1
[EINECS]
2090421
[Beilstein]
5-Amino-2-hydroxybenzoesäure
[German]
[ACD/IUPAC Name]
5-Amino-2-hydroxybenzoic acid
[ACD/IUPAC Name]
5-AS
5-ASA
89-57-6
[RN]
Acide 5-amino-2-hydroxybenzoïque
[French]
[ACD/IUPAC Name]
Asacolon
Benzoic acid, 5-amino-2-hydroxy-
[ACD/Index Name]
Canasa
[Trade name]
Lialda
[Trade name]
Mesacol
Mesalamine
[USP]
[USAN]
[Trade name]
Mesalazina
[Spanish]
Mesalazine
[Wiki] link-icon
[INN]
[BAN]
Mesalazinum
[Latin]
MFCD00007877
[MDL number]
Pentasa
[Trade name]
Rowasa
[Trade name]
Salofalk
[Trade name]
Salozinal
ZR CQ DVQ
[WLN]
SMILES
Nc1ccc(O)c(C(=O)O)c1
StdInChI
InChI=1S/C7H7NO3/c8-4-1-2-6(9)5(3-4)7(10)11/h1-3,9H,8H2,(H,10,11)
StdInChIKey
KBOPZPXVLCULAV-UHFFFAOYSA-N
Molecular Formula
C7H7NO3
Molecular Weight
153.137
EINECS
201-919-1
Beilstein Registry Number
2090421
MDL Number
MFCD00007877
Properties
Appearance
White powder
Melting Point
275-280 °C
Safety Data
Symbol
Signal Word
Warning
Hazard statements
H315,H317,H319,H335Precautionary Statements
P261 – P264 – P271 – P280 – P302 + P352 – P305 + P351 + P338Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
RIDADR
NONH for all modes of transport
WGK Germany
2
Specifications and Other Information of Our Mesalazine//Mesalamine//5-Aminosalicylic acid(5-ASA) CAS 89-57-6
Identification Methods
HNMR, HPLC
Standard
JP, CP , USP
Purity
99% min
Shelf Life
2 years
Storage
Under room temperature away from light
Productivity
1MT/month
Known Application
Mesalazine is the active component of sulfasalazine (SASP), responsible for its therapeutic effects in treating ulcerative colitis and Crohn’s disease. Its mechanism involves suppressing inflammation in the intestinal mucosa by targeting critical inflammatory pathways.
Treatment of Ulcerative Colitis : Effectively alleviates symptoms such as abdominal pain, diarrhea, and rectal bleeding.
Maintenance of Remission : Helps prevent relapses in patients with ulcerative colitis.
Management of Crohn’s Disease : Used for mild to moderate cases of Crohn’s disease as part of a comprehensive treatment plan.
Regulatory Approvals
Approved by China’s National Medical Products Administration (NMPA). Now is actively pursuing EU Certificate of Suitability (CEP) and Drug Master File (DMF) registration in Japan.
Links
This product is developed by our R&D company Caming Pharmaceutical Ltd (https://www.caming.com/).
Quick Inquiry
Fill out our inquiry form and one of our experts will be in touch with you shortly.

![Structure of Tetrachloro[2.2]paracyclophane <span class="caps">CAS</span> 30501-29-2 Structure of Tetrachloro[2.2]paracyclophane CAS 30501-29-2](https://www.watson-int.com/wp-content/uploads/2019/08/Structure-of-Tetrachloro2.2paracyclophane-CAS-30501-29-2.png)