Identification
CAS Number
7512-17-6
Name
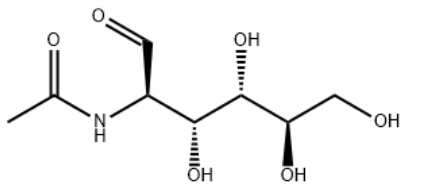
N-Acetyl-D-glucosamine
Synonyms
N-acetyl-α-D-glucosamine
2-(acetylamino)-2-deoxy-α-D-glucopyranose
233-115-1 [EINECS]
2-Acetamido-2-deoxy-a-D-glucopyranose
2-Acetamido-2-deoxy-α-D-glucopyranose [ACD/IUPAC Name]
2-acetamido-2-deoxy-α-δ-glucopyranose
2-Acetamido-2-desoxy-α-D-glucopyranose [German] [ACD/IUPAC Name]
2-Acétamido-2-désoxy-α-D-glucopyranose [French] [ACD/IUPAC Name]
2-acetylamino-2-deoxy-a-D-glucopyranose
2-acetylamino-2-deoxy-α-D-glucopyranose
7512-17-6 [RN]
Glucosamine, N-acetyl-
MFCD00064359 [MDL number]
N-((2S,3R,4R,5S,6R)-2,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-3-yl)acetamide
N-acetyl D-glucosamine
N-acetyl-a-D-glucosamine
N-ACETYL-D-GLUCOSAMINE
N-acetylglucosamine
N-acetyl-δ-glucosamine
T13TI5GH3D
α-D-GlcNAc
α-D-Glucopyranose, 2-(acetylamino)-2-deoxy- [ACD/Index Name]
α-GlcNAc
α-N-Acetyl-D-glucosamine
N;-acetyl-&α
N;-acetyl-&α;-D-glucosamine
??-D-GlcNAc
??-GlcNAc
[10036-64-3] [RN]
[6082-04-8] [RN]
2-(acetylamino)-2-deoxy-??-D-glucopyranose
2-(ACETYLAMINO)-2-DEOXY-A-D-GLUCOPYRANOSE
2-(acetylamino)-2-deoxy-A-D-glucopyranose ; 2-(acetylamino)-2-deoxy-??-D-glucopyranose ; N-acetyl-??-D-glucosamine ; ??-D-GlcNAc ; ??-GlcNAc
2-(acetylamino)-2-deoxy-D-glucose
2-acetamido-2-deoxy-α-d-glucopyranose
2-ACETAMIDO-2-DEOXY-α-D-GLUCOSE
6082-04-8 [RN]
95%
Acetyl Glucosamine
a-D-Glucopyranose,2-(acetylamino)-2-deoxy-
-D-glucosamine
EINECS 233-115-1
glucosamine, UDP-N-acetyl-
MAG
Methyl 2-acetamido-2-deoxy-a-D-glucopyranoside
Methyl-N-acetylglucosamine
MFCD00056076 [MDL number]
MFCD00136044 [MDL number]
N-[(2S,3R,4R,5S,6R)-2,4,5-trihydroxy-6-(hydroxymethyl)-3-tetrahydropyranyl]acetamide
N-[(2S,3R,4R,5S,6R)-2,4,5-trihydroxy-6-(hydroxymethyl)oxan-3-yl]acetamide
N-[(2S,3R,4R,5S,6R)-2,4,5-trihydroxy-6-(hydroxymethyl)oxan-3-yl]ethanamide
N-[(2S,3R,4R,5S,6R)-2,4,5-Trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-3-yl]acetamide
N-[(2S,3R,4R,5S,6R)-2,4,5-trihydroxy-6-(hydroxymethyl)tetrahydropyran-3-yl]acetamide
N-[(2S,3R,4R,5S,6R)-2,4,5-trihydroxy-6-methylol-tetrahydropyran-3-yl]acetamide
N-[(2S,5S,3R,4R,6R)-2,4,5-trihydroxy-6-(hydroxymethyl)-2H-3,4,5,6-tetrahydropy ran-3-yl]acetamide
N-[2,4,5-Trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-3-yl]acetamide
N-acetyl-??-D-glucosamine
N-Acetyl-D-glucosamine -2-H(3)
N-Acetyl-α-D-glucosamine
N-Acetyl-α-D-glucosaminide
NAG
NDG
PS-11249
QA-5909
SB 01966
SR-01000634877-1
UNII :T13TI5GH3D
UNII-T13TI5GH3D
WURCS=2.0/1,1,0/[a2122h-1a1-52*NCC/3=O]/1/
α-Methyl-N-Acetyl-D-Glucosamine
α-N-Acetyl-D-glucosamine, N-[(2S,3R,4R,5S,6R)-6-(Hydroxymethyl)-2,4,5-trihydroxytetrahydro-2H-pyran-3-yl]acetamide, 2-(Acetylamino)-2-deoxy-α-D-glucopyranose
α-N-Acetyl-D-glucosamine ; N-[(2S,3R,4R,5S,6R)-2,4,5-Trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-3-yl]acetamide
SMILES
CC(=O)N[C@@H]1[C@H]([C@@H]([C@H](O[C@@H]1O)CO)O)O
StdInChI
InChI=1S/C8H15NO6/c1-3(11)9-5-7(13)6(12)4(2-10)15-8(5)14/h4-8,10,12-14H,2H2,1H3,(H,9,11)/t4-,5-,6-,7-,8+/m1/s1
StdInChIKey
OVRNDRQMDRJTHS-PVFLNQBWSA-N
Molecular Formula
C8H15NO6
Molecular Weight
221.208
EINECS
231-368-2
MDL Number
MFCD00061615
Properties
Appearance
White powder
Safety Data
WGK Germany
3
MSDS Download
Specifications and Other Information of Our
Identification Methods
Assay
98%~102%
Odour
No odor
Particle Size
≥90% through 80 Mesh
Specific Rotation〔α〕d20
+39.0°~+43.0°
Ph(10mg/ml)
6~8
Chloride
≤0.1%
Loss On Drying
≤0.5%
Loss On Drying
≤0.5%
Residue On Ignition
≤0.1%
Iron
≤10ppm
Lead
≤10ppm
Arsenic
≤1.0ppm
Total Plate Count
≤1000CFU/g
Yeast & Mold
≤100CFU/g
Escherichia Coli
Not to be detected
Salmonella
Not to be detected
Shelf Life
2 years
Storage
Stored at room temperature for long time ; Sealed and keep away from light.
Known Application
N-Acetylglucosamine is a versatile ingredient widely used in pharmaceuticals, cosmetics, health supplements, and biomedical materials. Its primary sources include animal extraction and microbial fermentation.
In the pharmaceutical field, it helps improve joint health, promotes cartilage matrix synthesis by chondrocytes, enhances cartilage repair, and alleviates joint pain. As a derivative of glucosamine, it supports skin health and tissue repair.
In cosmetics, it stimulates the synthesis of hyaluronic acid (HA), enhancing skin hydration. It also repairs the skin barrier, reducing redness and sensitivity, while accelerating epidermal cell renewal and promoting wound healing.
General View of Documents
Links
This product is developed by our R&D company Ulcho Biochemical Limited (http://www.ulcho.com/).