Identification
CAS Number
50-35-1
Name
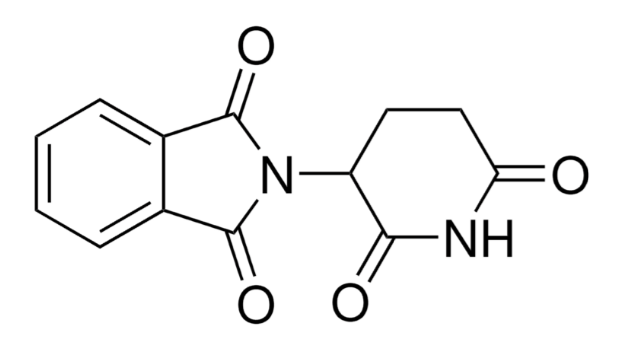
Thalidomide CAS 50-35-1
Synonyms
Thalidomide
(±)-2-(2,6-Dioxo-3-piperidinyl)-1H-isoindole-1,3(2H)-dione
(±)-Thalidomide
(±)-Thalidomide
1H-Isoindole-1,3(2H)-dione, 2-(2,6-dioxo-3-piperidinyl)- [ACD/Index Name]
2-(2,6-Dioxo-3-piperidinyl)-1H-isoindol-1,3(2H)-dion [German] [ACD/IUPAC Name]
2-(2,6-Dioxo-3-piperidinyl)-1H-isoindole-1,3(2H)-dione [ACD/IUPAC Name]
2-(2,6-Dioxo-3-pipéridinyl)-1H-isoindole-1,3(2H)-dione [French] [ACD/IUPAC Name]
2-(2,6-Dioxopiperidin-3-yl)-1H-isoindol-1,3(2H)-dion
2-(2,6-Dioxopiperidin-3-yl)-1H-isoindole-1,3(2H)-dione
2-(2,6-dioxopipéridin-3-yl)-1H-isoindole-1,3(2H)-dione
200-031-1 [EINECS]
4Z8R6ORS6L
50-35-1 [RN]
5-22-13-00224 [Beilstein]
Contergan [Wiki]
MFCD00153873 [MDL number]
Neosedyn
Neosydyn
Neurodyn
Softenon [Wiki]
Talidomida [Spanish] [INN]
Talidomide [DCIT]
thalidomide [French] [INN]
THALIDOMIDE, (R)-
THALIDOMIDE, (S)-
Thalidomidum [Latin] [INN]
Thalomide
TI4375000
UNII :4Z8R6ORS6L
талидомид [Russian] [INN]
ثاليدوميد [Arabic] [INN]
沙利度胺 [Chinese] [INN]
Telargan
(��)-Thalidomide
(±)-2-(2,6-Dioxo-3-piperidinyl)-1H-isoindole-1,3(2H)-dione
(±)-N-(2,6-dioxo-3-piperidyl)phthalimide
(±)-N-(2,6-Dioxo-3-piperidyl)phthalimide
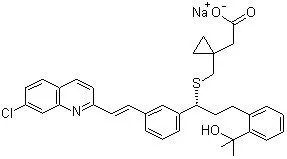
(±)-N-(2,6-dioxo-3-piperidyl)phthalimide ; (±)-thalidomide ; 1,3-dioxo-2-(2,6-dioxopiperidin-3-yl)isoindoline ; 2,6-dioxo-3-phthalimidopiperidine ; N-(2,6-dioxo-3-piperidyl)phthalimide ; N-Phthaloylglutamimide ; N-Phthalylglutamic acid imide ; Pro-ban M ; α-(N-phthalimido)glutarimide ; α-N-phthalylglutaramide ; α-phthalimidoglutarimide
(±)-Thalidomide
(±)-Thalidomide – CAS 50-35-1 – Calbiochem
(R)-(+)-Thalidomide
(R,S)-2-(2,6-dioxo-3-piperidinyl)-1H-isoindole-1,3(2H)-dione
(S)-(−)-Thalidomide
1,3-Dioxo-2-(2,6-dioxopiperidin-3-yl)isoindoline
1219177-18-0 [RN]
14088-68-7 [RN]
1H-Isoindole-1,3(2H)-dione, 2-(2,6-dioxo-3-piperidinyl)-, (R)-
1H-Isoindole-1,3(2H)-dione, 2-(2,6-dioxo-3-piperidinyl)-, (S)-
2-(2,6-dioxo-3-piperidyl)isoindoline-1,3-dione
2-(2,6-dioxoazaperhydroin-3-yl)benzo[c]azoline-1,3-dione
2-(2,6-dioxopiperidin-3-yl)-2,3-dihydro-1H-isoindole-1,3-dione
2-(2,6-dioxopiperidin-3-yl)isoindole-1,3-dione
2-(2,6-Dioxo-piperidin-3-yl)-isoindole-1,3-dione
2-(2,6-dioxopiperidin-3-yl)isoindoline-1,3-dione
2-(6-hydroxy-2-oxo-2,3,4,5-tetrahydropyridin-3-yl)-2,3-dihydro-1H-isoindole-1,3-dione
2,6-Dioxo-3-phthalimidopiperidine
2-[(3S)-2,6-dioxo-3-piperidinyl]isoindole-1,3-dione
200-031-1MFCD00153873
2614-06-4 [RN]
2-phthalimidoglutarimide
3-Phthalimidoglutarimide
6160-65-2 [RN]
731-40-8 [RN]
Actimid
Algosediv
a-Phthalimidoglutarimide
Asidon 3
Asmadion
Asmaval
Bonbrain
Calmore
Calmorex
Contergan (Trade name)
Contergan ; Distaval ; K-17 ; Sedalis ; Softenon ; Talimol ; Thaled
Corronarobetin
D013792
Distaval
Distaval (Trade name)
Distaval ; K-17 ; Sedalis ; Softenon ; Talimol
Distaxal
Distoval
Ectiluran
Enterosediv
Gastrinide
Glupan
Glutanon
Glutarimide, 2-phthalimido-
Grippex
Hippuzon
https://www.ebi.ac.uk/chembl/compoundreportcard/CHEMBL468/
Imida-lab
Imidan
Imidan (peyta)
Imidene
Isomin
K-17 (Trade name)
Kedavon
Kevadon
l-Thalidomide
Lulamin
N-(2,6-dioxo-3-piperidinyl)phthalimide
N-(2,6-Dioxo-3-piperidyl)phthalimide
N-(2,6-Dioxopiperidin-3-yl)phthalimide
Neaufatin
NEO
Nerosedyn
Neufatin
Neurosedin
Neurosedym
Neurosedyn
Nevrodyn
Nibrol
Noctosediv
Noxodyn
N-Phthalimidoglutamic acid imide
N-Phthaloylglutamimide
N-Phthalylglutamic acid imide
N-Phthalyl-glutaminsaeure-imid [German]
N-Phthalyl-glutaminsaeure-imid
OR-1446
Pangul
Pantosediv
Pharmakon1600-01503607
Pharmion
Phthalimide, N-(2,6-dioxo-3-piperidyl)-
Pituitary Adenylate Cyclase-Activating Polypeptide 1-38 |
Predni-sediv
Pro-Bam M
Pro-ban M
Profarmil
Psycholiquid
Psychotablets
Quetimid
Quietoplex
rac-2-(2,6-dioxopiperidin-3-yl)-1H-isoindole-1,3(2H)-dione
Sandormin
Sedalis
Sedalis (Trade name)
Sedalis sedi-lab
Sedimide
Sedin
Sedisperil
Sedoval
Shin-naito S
Shinnibrol
Sleepan
Slipro
Softenil
Softenon (Trade name)
Suaramide
Synovir
Talargan
talidomida
talidomida ; thalidomide ; thalidomidum
TALIMOL
Talimol (Trade name)
Talinol
Talismol
Talizer
Telagan
Telargean
Tensival
Thaled
Thaled (TN)
Thalidomide (JAN/USP/INN)
Thalidomide;N-(2,6-Dioxopiperidin-3-yl)phthalimide
thalidomidum
Thalin
Thalinette
Thalomid [Wiki]
Theophilcholine
Ulcerfen [Trade name]
Valgis
Valgraine
WLN : T56 BVNVJ C- DT6VMVTJ
WLN : T56 BVNVJ C- DT6VMVTJ -D
WLN : T56 BVNVJ C- DT6VMVTJ -L
Yodomin
α-(N-phthalimido)glutarimide
α-(N-Phthalimido)glutarimide
α-(N-Phthalimido)glutarimide
α-N-phthalylglutaramide
α-N-Phthalylglutaramide
α-N-Phthalylglutaramide
α-phthalimidoglutarimide
α-Phthalimidoglutarimide
α-Phthalimidoglutarimide
талидомид
ثاليدوميد
沙利度胺
SMILES
c1ccc2c(c1)C(=O)N(C2=O)C3CCC(=NC3=O)O
StdInChI
InChI=1S/C13H10N2O4/c16-10-6-5-9(11(17)14-10)15-12(18)7-3-1-2-4-8(7)13(15)19/h1-4,9H,5-6H2,(H,14,16,17)
StdInChIKey
UEJJHQNACJXSKW-UHFFFAOYSA-N
Molecular Formula
C13H10N2O4
Molecular Weight
258.229
EINECS
200-031-1
Beilstein Registry Number
5-22-13-00224
MDL Number
MFCD00153873
Properties
Appearance
White powder
Safety Data
RIDADR
NONH for all modes of transport
Specifications and Other Information of Our Thalidomide CAS 50-35-1
Identification Methods
HNMR, HPLC
Purity
95% min
Storage
Store at 2-8℃ for long time.
Features
Substrate Proteins : The primary ligands for E3 ligases are substrate proteins that need to be ubiquitinated for degradation or regulation. These substrate proteins can vary widely depending on the specific E3 ligase and the cellular process it regulates.
Ubiquitin : In the ubiquitination process, ubiquitin itself acts as a ligand. It forms a thioester bond with the active site cysteine of the E3 ligase before being transferred to the substrate protein.
Adaptor Proteins : Some E3 ligases require adaptor proteins to facilitate substrate recognition and ubiquitination. These adaptor proteins can also act as ligands for the E3 ligase.
Small Molecules : In some cases, small molecules or chemical compounds can modulate the activity of E3 ligases by binding to them directly or affecting their interactions with other proteins.
Post-translational Modifications (PTMs): PTMs of either the E3 ligase itself or its substrate proteins can also serve as ligands, regulating the ubiquitination process.
Overall, the ligands for E3 ligases play crucial roles in substrate recognition, ubiquitin transfer, and the regulation of cellular processes.
Known Application
Substrate Recognition : Ligands facilitate the recognition of specific substrate proteins by E3 ligases. These ligands can be protein motifs, post-translational modifications (such as phosphorylation or acetylation), or adaptor proteins that bring substrates to the E3 ligase.
Ubiquitination : Once a substrate is recognized, E3 ligases catalyze the transfer of ubiquitin molecules to the substrate protein. These ligands are crucial for facilitating the transfer of ubiquitin from the E2 ubiquitin-conjugating enzyme to the substrate.
Regulation of Protein Stability : Ubiquitination mediated by E3 ligases marks substrate proteins for degradation by the proteasome. Ligands for E3 ligases thus regulate the stability of proteins in the cell by targeting them for degradation.
Regulation of Protein Function : Ubiquitination can also regulate the activity, localization, or interaction partners of substrate proteins without leading to degradation. Ligands for E3 ligases are involved in this regulatory process by controlling the extent and site of ubiquitination.
Cellular Signaling : Ubiquitination mediated by E3 ligases is involved in various cellular signaling pathways, including those regulating cell cycle progression, DNA repair, immune response, and apoptosis. Ligands for E3 ligases modulate these signaling pathways by targeting specific proteins for ubiquitination.
Overall, ligands for E3 ligases are essential for substrate recognition, ubiquitination, and the regulation of protein stability and function, thereby influencing numerous cellular processes and signaling pathways.
Links
This product is developed by our R&D company Caming Pharmaceutical Ltd (https://www.caming.com/).
Quick Inquiry
Fill out our inquiry form and one of our experts will be in touch with you shortly.