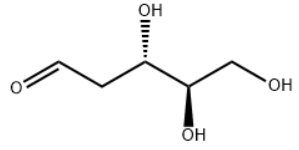
Identification
CAS Number
533-67-5
Name
2-DEOXY-D-ARABINOSE
Synonyms
(3S,4R)-3,4,5-Trihydroxypentanal
1724-14-7
[RN]
2-Deoxy-D-arabinose
2-Deoxy-D-erythro-pentose
[IUPAC name – generated by ACD/Name]
2-Deoxy-D-erythropentose
2-Deoxy-D-Ribose
2-Deoxy-β-D-erythro-pentose
2-deoxyribose
2-Desoxy-D-erythro-pentose
[German]
[IUPAC name – generated by ACD/Name]
2-Désoxy-D-érythro-pentose
[French]
[IUPAC name – generated by ACD/Name]
217-028-6
[EINECS]
533-67-5
[RN]
D-2-Deoxyarabinose
D-2-Deoxyribose
D-2-RIBODESOSE
D-erylhro-2-Deoxypentose
D-erythro-Pentose, 2-deoxy-
[IUPAC index name – generated by ACD/Name]
D-Ribose, 2-deoxy-
Deoxyribose
[Wiki]
desoxyribose
LSW4H01241
[UNII]
Thyminose
SMILES
O=CCC@HC@HCO
StdInChI
InChI=1S/C5H10O4/c6-2-1-4(8)5(9)3-7/h2,4-5,7-9H,1,3H2/t4-,5+/m0/s1
StdInChIKey
ASJSAQIRZKANQN-CRCLSJGQSA-N
Molecular Formula
C5H10O4
Molecular Weight
134.131
EINECS
208-573-0
Beilstein Registry Number
1721978
MDL Number
MFCD00135904
Properties
Appearance
White to off-white crystalline powder
Safety Data
Symbol
Signal Word
WGK Germany
3
MSDS Download
Specifications and Other Information of Our 2-DEOXY-D-ARABINOSE CAS 533-67-5
Identification Methods
HPLC
Purity
98% min
Rotation
-54° ~ -60°
Heavy Metals
≤10ppm
PH
6.0 ~8.0
Loss on Drying
≤1.0%
Residue on Ignition
≤0.5%
Shelf Life
2 years
Storage
Under room temperature away from light
Known Application
2-Deoxy-D-ribose is widely used in DNA research, cell oxidative stress experiments, biochemical analysis, and nucleoside synthesis. It is not a product with direct industrial or pharmaceutical applications. Main Applications 1. Research Uses As a basic building block of DNA, it is often used in molecular biology research. The metabolism and role of 2-deoxy-D-ribose are often explored in studies of DNA damage and repair mechanisms. 2. Biochemical Reagents It is often used as a reducing sugar in biochemical experiments, such as testing redox reactions and free radical reactions. It is used in detection and quantification methods (for example, determining the content of deoxysugars). 3. Medical and Pharmacological Research It is used as an inducing factor in cell oxidative stress and apoptosis experiments because it promotes the generation of reactive oxygen species (ROS). Studies have used it to investigate the mechanisms of oxidative stress associated with cardiovascular disease, diabetic complications, and other conditions. 4. Synthetic Applications It can serve as an intermediate in the synthesis of certain nucleosides, nucleotides, or nucleoside analogs.
General View of Documents
Links
This product is developed by our R&D company Caming Pharmaceutical Limited (http://www.caming.com/).
Quick Inquiry
Fill out our inquiry form and one of our experts will be in touch with you shortly.

![Structure of 1,2-Dihydrocyclobuta[a]naphthalene <span class="caps">CAS</span> 32277-35-3 Structure of 1,2-Dihydrocyclobuta[a]naphthalene CAS 32277-35-3](https://www.watson-int.com/wp-content/uploads/2016/04/Structure-of-12-Dihydrocyclobutaanaphthalene-CAS-32277-35-3.png)