Identification
CAS Number
6949-73-1
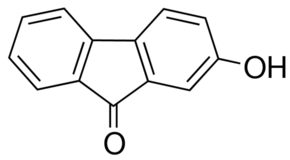
Name
2-Hydroxy-9-fluorenone
Synonyms
230-119-5 [EINECS]
2-Hydroxy-9-fluorenone
2-Hydroxy-9H-fluoren-9-on [German] [ACD/IUPAC Name]
2-Hydroxy-9H-fluoren-9-one [ACD/IUPAC Name]
2-Hydroxy-9H-fluorén-9-one [French] [ACD/IUPAC Name]
6949-73-1 [RN]
9H-Fluoren-9-one, 2-hydroxy- [ACD/Index Name]
(1-(2-(Dimethylamino)ethyl)-1H-pyrazol-4-yl)boronic acid
[6949-73-1]
2-[(3-chloroanilino)methylidene]propanedinitrile
2-hydroxy-9-fluorenone, 97%
2-HYDROXY-9-FLUORENONE|2-HYDROXY-9H-FLUOREN-9-ONE
2-hydroxyfluoren-9-one
‘6949-73-1
9H-Fluoren-9-one,2-hydroxy-
fluoren-9-one, 2-hydroxy-
http:////www.amadischem.com/proen/607340/
SMILES
c1ccc2c(c1)-c3ccc(cc3C2=O)O
StdInChI
InChI=1S/C13H8O2/c14-8-5-6-10-9-3-1-2-4-11(9)13(15)12(10)7-8/h1-7,14H
StdInChIKey
GXUBPHMYNSICJC-UHFFFAOYSA-N
Molecular Formula
C13H8O2
Molecular Weight
196.201
EINECS
230-119-5
MDL Number
MFCD00001155
Properties
Appearance
Yellow to orange powder
Melting Point
204-206 °C(lit.)
Safety Data
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
RIDADR
NONH for all modes of transport
Specifications and Other Information of Our 2-Hydroxy-9-fluorenone CAS 6949-73-1
Identification Methods
HNMR, HPLC
Purity
98% min
Shelf Life
2 years
Storage
Under room temperature away from light
Known Application
Fluorenone and its derivatives have become the main monomer or modifier of a new type of engineering plastics. Some foreign companies have developed such products and put them on the market. At present, 2,7-dihydroxy-9-fluorenone, as an important pharmaceutical intermediate and material functional monomer, can be used in the pharmaceutical industry to synthesize anti-tumor, sympathetic nerve inhibitors, etc.; in agriculture, it can be used to synthesize herbicides and Insecticides, etc.; in the plastics industry, it can be used to synthesize bisphenol products as stabilizers and plasticizers in the polymerization process and to prepare functional polymer materials ; in the field of liquid crystal materials, it can be used to synthesize materials that are prone to mesogenic phenomena New organic compounds.