Identification
CAS Number
78512-32-0
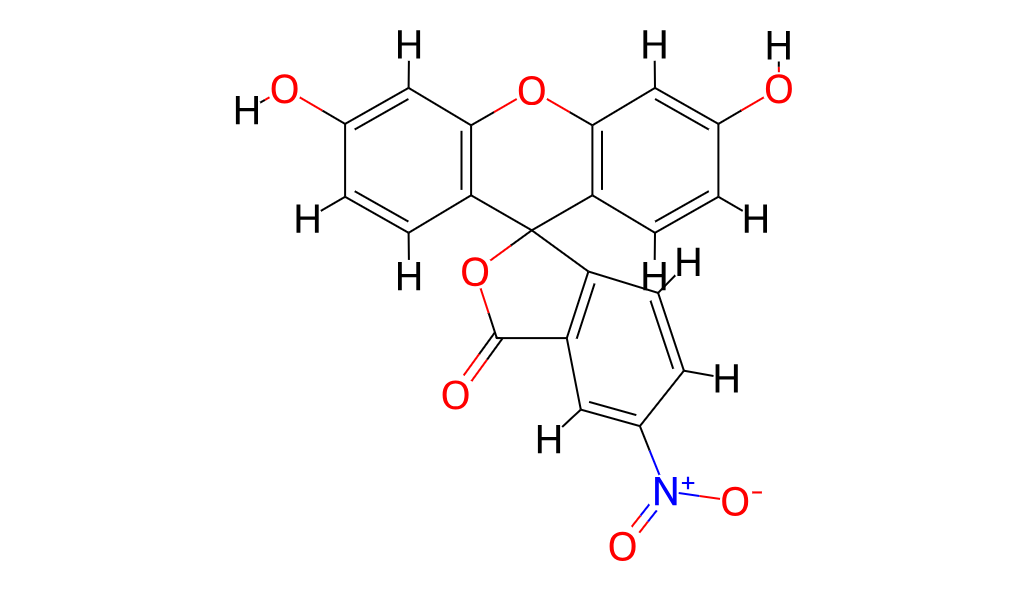
Name
5(6)-Nitrofluorescein
Synonyms
3′,6′-Dihydroxy-5-nitro-3H-spiro[2-benzofuran-1,9′-xanthen]-3-on
3′,6′-Dihydroxy-5-nitro-3H-spiro[2-benzofuran-1,9′-xanthen]-3-one
3′,6′-Dihydroxy-5-nitro-3H-spiro[2-benzofuran-1,9′-xanthen]-3-one[French][ACD/IUPAC Name]
3′,6′-dihydroxy-5-nitro-3H-spiro[isobenzofuran-1,9′-xanthen]-3-one
Spiro(isobenzofuran-1(3H),9′-(9H)xanthen)-3-one, 3′,6′-dihydroxy-5-nitro-
Spiro[isobenzofuran-1(3H),9′-[9H]xanthen]-3-one, 3′,6′-dihydroxy-5-nitro-
3′,6′-dihydroxy-5-nitrospiro[2-benzofuran-1,9′-xanthen]-3-one
3′,6′-DIHYDROXY-5-NITROSPIRO[2-BENZOFURAN-1,9′-XANTHENE]-3-ONE
3′,6′-dihydroxy-6-nitrospiro[2-benzofuran-3,9′-xanthene]-1-one
4-NITROFLUORESCEIN
5-Nitrofluorescein
5-Nitrofluorescein (isomer I)
MFCD00059693[MDL number]
MFCD00059694[MDL number]
MFCD00135108[MDL number]
Nitrofluorescein, isomer 1
Spiro[isobenzofuran-1(3H),9′-[9H]xanthen]-3-one,3′,6′-dihydroxy-5-nitro-
SMILES
O=C1OC2(c3ccc(O)cc3Oc3cc(O)ccc32)c2ccc(N+[O-])cc21
StdInChI
InChI=1S/C20H11NO7/c22-11-2-5-15-17(8-11)27-18-9-12(23)3-6-16(18)20(15)14-4-1-10(21(25)26)7-13(14)19(24)28-20/h1-9,22-23H
StdInChIKey
UACQOMKZFGNRBL-UHFFFAOYSA-N
Molecular Formula
C20H11NO7
Molecular Weight
377.308
MDL Number
MFCD00059693
Properties
Appearance
Yellow solid
Safety Data
RIDADR
NONH for all modes of transport
Specifications and Other Information of Our 5(6)-Nitrofluorescein CAS 78512-32-0
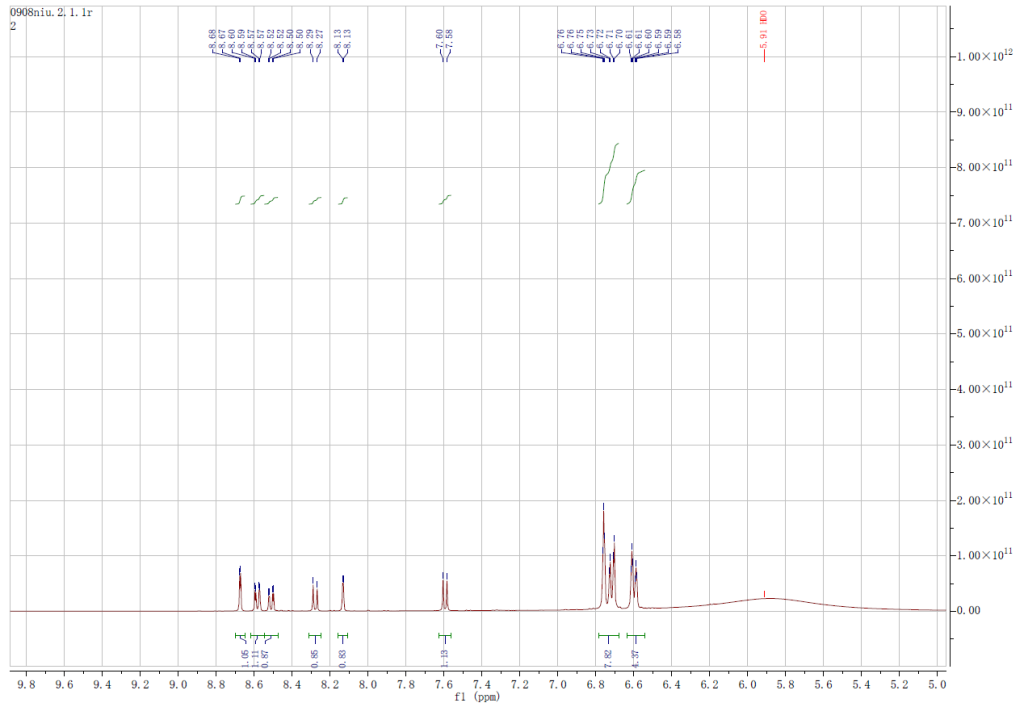
Identification Methods
HNMR, HPLC
Purity
97% min
Shelf Life
1 year
Storage
Under room temperature away from light
Known Application
- Fluorescent probe development
Due to its adjustable fluorescence response at specific wavelengths, 5(6)-Nitrofluorescein is often used to develop fluorescent probes and molecular markers, especially in bioimaging or analytical chemistry. Nitro substitution will quench fluorescence and can be used to construct “on/off” probes (such as indicators for enzyme reaction monitoring or redox reactions). - Biolabeling / covalent coupling reaction
It can react with amino compounds through its carboxyl group to form a stable covalent bond, which is used for labeling and tracking biological macromolecules such as proteins and nucleic acids. It is used in conjunction with fluorescence microscopy, flow cytometry, etc. - Spectroscopic research
It is used as a model compound to study spectral properties such as intermolecular energy transfer, electronic effects, and changes in fluorescence quantum yield. - Used to synthesize more complex fluorescent dyes or sensors
It is an intermediate or starting material for many fluorescent derivatives (such as FRET probes, reactive oxygen detection dyes, etc.).
General View of Documents


Links
This product is developed by our R&D company Watson Bio Ltd (https://www.watson-bio.com/).
Quick Inquiry
Fill out our inquiry form and one of our experts will be in touch with you shortly.