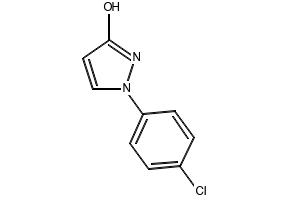
![Strcuture of 6-Chloro-1H-pyrazolo[4,3-c]pyridine <span class="caps">CAS</span> 1206979-33-0 Strcuture of 6-Chloro-1H-pyrazolo[4,3-c]pyridine CAS 1206979-33-0](https://www.watson-int.com/wp-content/uploads/2025/04/Strcuture-of-6-Chloro-1H-pyrazolo43-cpyridine-CAS-1206979-33-0.png)
Identification
CAS Number
1206979-33-0
Name
6-Chloro-1H-pyrazolo[4,3-c]pyridine
Synonyms
1206979-33-0[RN]
1H-Pyrazolo[4,3-c]pyridine, 6-chloro-
[ACD/Index Name]
6-Chlor-1H-pyrazolo[4,3-c]pyridin
[German]
[ACD/IUPAC Name]
6-chloro-1H-pyrazolo[4,3-c]pyridine
[ACD/IUPAC Name]
6-Chloro-1H-pyrazolo[4,3-c]pyridine
[French]
[ACD/IUPAC Name]
MFCD17010105
[MDL number]
Unverified
2-(Amino(phenyl)methylene)malononitrile
20300-02-1
[RN]
4-Chloro-1H-pyrazolo[4,3-c]pyridine
[ACD/IUPAC Name]
4649-09-6
[RN]
6-chloro-1h-pyrazolo4,3-cpyridine
6-chloro-1h-pyrazolo[4,3-c]pyridine(wxc01805)
6-Chloro-1H-pyrazolo[4,3-c]pyridine,
6-chloro-2H-pyrazolo[4,3-c]pyridine
6-Chloro-5-azaindazole
871836-51-0
[RN]
95%
AGN-PC-0BMJND
CTK8B5457
MolPort-023-219-348
PS-J-068
Thiophene-2-thiocarboxamide
SMILES
Clc1cc2[nH]ncc2cn1
StdInChI
InChI=1S/C6H4ClN3/c7-6-1-5-4(2-8-6)3-9-10-5/h1-3H,(H,9,10)
StdInChIKey
AAJIQIWPVIWCGA-UHFFFAOYSA-N
Molecular Formula
C6H4ClN3
Molecular Weight
153.569
MDL Number
MFCD17010105
Properties
Appearance
Light yellow to brown solid
Solubility
Soluble in ethanol
Safety Data
RIDADR
NONH for all modes of transport
WGK Germany
3
Specifications and Other Information of Our 6-Chloro-1H-pyrazolo[4,3-c]pyridine CAS 1206979-33-0
Identification Methods
HNMR, HPLC
Purity
98% min
Shelf Life
2 years
Storage
Under room temperature away from light
Known Application
- Pharmaceutical intermediates : This is its most common use. This type of structure is widely used to synthesize targeted kinase drugs (such as PI3K, JAK, BTK inhibitors) or central nervous system drugs. For example, it is used as a core skeleton in the synthesis route of anti-cancer, anti-inflammatory, antiviral and neurological disease drugs.
- Drug lead compound design : Pyrazolopyridine structure is valued in medicinal chemistry for its biological activity. The chlorine atom site of this compound can be used for further functional group introduction reactions (such as Suzuki or Buchwald coupling reactions) to construct complex molecules.
- Scientific research use : Commonly used in high-throughput screening (HTS) or structure-activity relationship studies (SAR) in the new drug development stage.
General View of Documents
![HPLC of 6-Chloro-1H-pyrazolo[4,3-c]pyridine CAS 1206979-33-0](https://www.watson-int.com/wp-content/uploads/2025/04/HPLC-of-6-Chloro-1H-pyrazolo43-cpyridine-CAS-1206979-33-0-1.png)
![HPLC of 6-Chloro-1H-pyrazolo[4,3-c]pyridine CAS 1206979-33-0](https://www.watson-int.com/wp-content/uploads/2025/04/HPLC-of-6-Chloro-1H-pyrazolo43-cpyridine-CAS-1206979-33-0-1.png)
![HPLC of 6-Chloro-1H-pyrazolo[4,3-c]pyridine CAS 1206979-33-0](https://www.watson-int.com/wp-content/uploads/2025/04/HPLC-of-6-Chloro-1H-pyrazolo43-cpyridine-CAS-1206979-33-0-1.png)
![1HNMR of 6-Chloro-1H-pyrazolo[4,3-c]pyridine CAS 1206979-33-0](https://www.watson-int.com/wp-content/uploads/2025/04/1HNMR-of-6-Chloro-1H-pyrazolo43-cpyridine-CAS-1206979-33-0-1024x687.png)
![1HNMR of 6-Chloro-1H-pyrazolo[4,3-c]pyridine CAS 1206979-33-0](https://www.watson-int.com/wp-content/uploads/2025/04/1HNMR-of-6-Chloro-1H-pyrazolo43-cpyridine-CAS-1206979-33-0-1024x687.png)
![1HNMR of 6-Chloro-1H-pyrazolo[4,3-c]pyridine CAS 1206979-33-0](https://www.watson-int.com/wp-content/uploads/2025/04/1HNMR-of-6-Chloro-1H-pyrazolo43-cpyridine-CAS-1206979-33-0-1024x687.png)
![HNMR of 6-Chloro-1H-pyrazolo[4,3-c]pyridine CAS 1206979-33-0](https://www.watson-int.com/wp-content/uploads/2025/04/HNMR-of-6-Chloro-1H-pyrazolo43-cpyridine-CAS-1206979-33-0-1024x686.png)
![HNMR of 6-Chloro-1H-pyrazolo[4,3-c]pyridine CAS 1206979-33-0](https://www.watson-int.com/wp-content/uploads/2025/04/HNMR-of-6-Chloro-1H-pyrazolo43-cpyridine-CAS-1206979-33-0-1024x686.png)
![HNMR of 6-Chloro-1H-pyrazolo[4,3-c]pyridine CAS 1206979-33-0](https://www.watson-int.com/wp-content/uploads/2025/04/HNMR-of-6-Chloro-1H-pyrazolo43-cpyridine-CAS-1206979-33-0-1024x686.png)
![HNMR of 6-Chloro-1H-pyrazolo[4,3-c]pyridine CAS 1206979-33-0](https://www.watson-int.com/wp-content/uploads/2025/04/HNMR-of-6-Chloro-1H-pyrazolo43-cpyridine-CAS-1206979-33-0-1-1024x668.png)
![HNMR of 6-Chloro-1H-pyrazolo[4,3-c]pyridine CAS 1206979-33-0](https://www.watson-int.com/wp-content/uploads/2025/04/HNMR-of-6-Chloro-1H-pyrazolo43-cpyridine-CAS-1206979-33-0-1-1024x668.png)
![HNMR of 6-Chloro-1H-pyrazolo[4,3-c]pyridine CAS 1206979-33-0](https://www.watson-int.com/wp-content/uploads/2025/04/HNMR-of-6-Chloro-1H-pyrazolo43-cpyridine-CAS-1206979-33-0-1-1024x668.png)
Links
This product is developed by our R&D company Caming Pharmaceutical Ltd (https://www.caming.com/).
Quick Inquiry
Fill out our inquiry form and one of our experts will be in touch with you shortly.

![Structure of 1,3-bis[3-(dimethylamino)propyl]urea <span class="caps">CAS</span> 52338-87-1 Structure of 1,3-bis[3-(dimethylamino)propyl]urea CAS 52338-87-1](https://www.watson-int.com/wp-content/uploads/2024/04/Structure-of-13-bis3-dimethylaminopropylurea-CAS-52338-87-1.png)
![Structure of <span class="caps">TMBI1</span>,1,2-Trimethyl-1H-benz[e]indole <span class="caps">CAS</span> 41532-84-7 Structure of TMBI1,1,2-Trimethyl-1H-benz[e]indole CAS 41532-84-7](https://www.watson-int.com/wp-content/uploads/2014/12/Structure-of-TMBI112-Trimethyl-1H-benzeindole-CAS-41532-84-7.png)