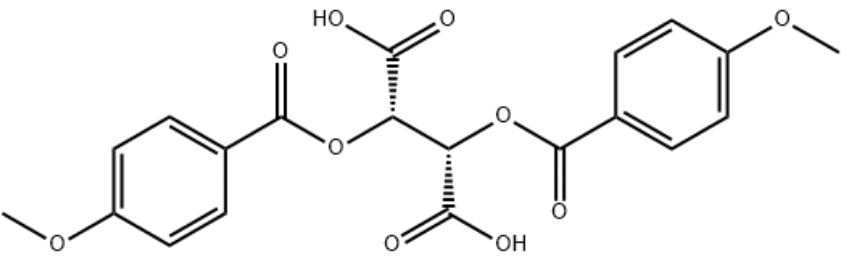
Identification
CAS Number
191605-10-4
Name
Di-p-anisoyl-D-tartaric acid CAS 191605-10-4
Synonyms
(2S,3S)-2,3-bis(4-methoxybenzoyloxy)butanedioic acid
(2S,3S)-2,3-Bis[(4-methoxybenzoyl)oxy]bernsteinsäure [German] [ACD/IUPAC Name]
(2S,3S)-2,3-Bis[(4-methoxybenzoyl)oxy]butanedioic acid
(2S,3S)-2,3-Bis[(4-methoxybenzoyl)oxy]succinic acid [ACD/IUPAC Name]
(2S,3S)-2,3-Bis{[(4-methoxyphenyl)carbonyl]oxy}butanedioic acid
(S,S)-Di-p-anisoyltartaric acid
191605-10-4 [RN]
1OR DVOYVQYVQOVR DO1 &&(2S,3S)- Form [WLN]
3227682 [Beilstein]
Acide (2S,3S)-2,3-bis[(4-méthoxybenzoyl)oxy]succinique [French] [ACD/IUPAC Name]
Butanedioic acid, 2,3-bis[(4-methoxybenzoyl)oxy]-, (2S,3S)- [ACD/Index Name]
Di-p-anisoyl-D-tartaric acid [ACD/IUPAC Name]
(2S,3S)-2,3-bis(4-methoxyphenylcarbonyloxy)butanedioic acid
(2S,3S)-2,3-bis[(4-methoxyphenyl)carbonyloxy]butanedioic acid
[191605-10-4] [RN]
98%
Jsp003934
MFCD07368366 [MDL number]
O,O’-Di-p-anisoyl-D-tartaric acid
OR-4444
PubChem6043
SCHEMBL935315
SMILES
COC1=CC=C(C=C1)C(=O)O[C@@H]([C@@H](C(=O)O)OC(=O)C2=CC=C(C=C2)OC)C(=O)O
StdInChI
InChI=1S/C20H18O10/c1-27-13-7-3-11(4-8-13)19(25)29-15(17(21)22)16(18(23)24)30-20(26)12-5-9-14(28-2)10-6-12/h3-10,15-16H,1-2H3,(H,21,22)(H,23,24)/t15-,16-/m0/s1
StdInChIKey
KWWCVCFQHGKOMI-HOTGVXAUSA-N
Molecular Formula
C20H18O10
Molecular Weight
418.351
MDL Number
MFCD07368366
Properties
Appearance
White Powder
Safety Data
Symbol
Signal Word
Warning
Hazard statements
H302, H315, H319, H332, H335Precautionary Statements
P261, P280, P305, P338, P351WGK Germany
3
MSDS Download
Specifications and Other Information of Our Di-p-anisoyl-D-tartaric acid CAS 191605-10-4
Purity
98% min
Specific Rotation
+165° ~ +171°
Water
≤0.5%
Shelf Lif
2 years
Storage
Store at room temperature, in container tightly sealed ; Protect from light.
Known Application
D-(+)-Di-p-methoxybenzoyl-tartaric acid (DPMTA) is a chiral compound, is commonly used in organic chemistry and pharmaceutical chemistry as a chiral resolving agent to separate racemic mixtures. It can form diastereomeric salts with racemic compounds, which can then be separated by crystallization or chromatography techniques. As a chiral catalyst or chiral ligand, DPMTA is used in asymmetric synthesis reactions to enhance enantioselectivity. This is particularly important in the synthesis of chiral drugs or chiral materials.
Links
This product is developed by our R&D company Caming Pharmaceutical Limited (http://www.caming.com/).
Quick Inquiry
Fill out our inquiry form and one of our experts will be in touch with you shortly.

![Structure of 4,8-bis(5-(2-ethylhexyl)thieno[3,2-b]thiophene-2-yl)benzo[1,2-b4,5-b’]dithiophene <span class="caps">CAS</span> 1494614-30-0 Structure of 4,8-bis(5-(2-ethylhexyl)thieno[3,2-b]thiophene-2-yl)benzo[1,2-b4,5-b’]dithiophene CAS 1494614-30-0](https://www.watson-int.com/wp-content/uploads/2018/06/Structure-of-48-bis5-2-ethylhexylthieno32-bthiophene-2-ylbenzo12-b45-b’dithiophene-CAS-1494614-30-0.png)