Identification
CAS Number
37943-90-1
Name
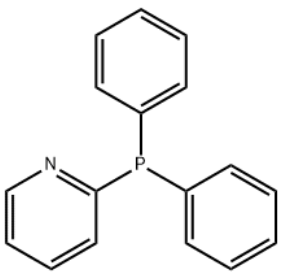
Diphenyl-2-pyridylphosphine
Synonyms
2-(Diphenylphosphino)pyridin [German] [ACD/IUPAC Name]
2-(Diphenylphosphino)pyridine [ACD/IUPAC Name]
2-(Diphénylphosphino)pyridine [French] [ACD/IUPAC Name]
2-diphenylphosphanyl-pyridine
2-diphenylphosphinopyridine
2-pyridyldiphenylphosphine
37943-90-1 [RN]
diphenyl(2-pyridinyl)phosphine
Diphenyl-2-pyridylphosphine [Wiki]
MFCD00192108 [MDL number]
Pyridine, 2-(diphenylphosphino)- [ACD/Index Name]
2-(diphenylphosphanyl)pyridine
2-(Diphenylphosphino)pyridine|2-(Diphenylphosphanyl)pyridine
98%
bisphenyl-(pyridin-2-yl)phosphine
Diphenyl 2-pyridyl phosphine
diphenyl(2-pyridyl)phosphine
diphenyl(pyridin-2-yl)phosphane
diphenylphosphino-pyridine
diphenylpyridinylphosphine
DPPPY
OR-0960
SMILES
c1ccc(cc1)P(c2ccccc2)c3ccccn3
StdInChI
InChI=1S/C17H14NP/c1-3-9-15(10-4-1)19(16-11-5-2-6-12-16)17-13-7-8-14-18-17/h1-14H
StdInChIKey
SVABQOITNJTVNJ-UHFFFAOYSA-N
Molecular Formula
C17H14NP
Molecular Weight
263.273
MDL Number
MFCD00192108
Properties
Appearance
White to Off-white crystalline powder
Safety Data
Symbol
Signal Word
Warning
Hazard statements
H315,H319,H335,H413Precautionary Statements
P261 – P264 – P271 – P273 – P302 + P352 – P305 + P351 + P338WGK Germany
3
MSDS Download
Specifications and Other Information of Our Diphenyl-2-pyridylphosphine CAS 37943-90-1
Identification Methods
HNMR, HPLC
Purity
99% min
Melting Point
82~83℃
Shelf Life
1 year
Storage
Slowly oxidized in the air for a long time, vacuum pack, and store in cold storage
Known Application
Ligand in metal-catalyzed reactions :
Carbonylation : Facilitates the introduction of carbonyl groups into organic molecules.
Hydration : Aids in the addition of water to unsaturated compounds, forming alcohols or other functionalized derivatives.
Dehydrogenative coupling : Promotes coupling reactions by removing hydrogen, often forming C-C or C-N bonds.
Carbostannylation : Catalyzes the introduction of stannyl (Sn) groups into organic compounds.
Dimethylstannylation : Specific variant of stannylation, introducing dimethylstannyl groups.
Silylation : Facilitates the introduction of silyl groups, improving stability and reactivity of substrates.
Reagent in Mitsunobu reactions :
Used in the conversion of alcohols to various functional groups, such as esters, ethers, or amines, under mild conditions.
General View of Documents
Links
This product is developed by our R&D company Warshel Chemical Ltd (https://www.warshel.com/).
Quick Inquiry
Fill out our inquiry form and one of our experts will be in touch with you shortly.