Semaglutide, a groundbreaking product in the GLP-1 drug class, owes its extended half-life and enhanced receptor affinity largely to its unique side chain, Ste-Glu-AEEA-AEEA-OSU (CAS : 1169630-40-3). This side chain covalently modifies the peptide backbone, significantly improving pharmacokinetics and therapeutic performance. However, its complex structure presents two critical synthetic challenges :



- Precise Assembly of Repetitive AEEA Units :
The side chain features consecutive AEEA (aminoethoxyethoxyacetic acid) units, which require stepwise coupling via highly activated intermediates (e.g., AEEA-AEEA). Any impurities or deviations compromise downstream reaction efficiency and may trigger irreversible byproducts. - Stereochemistry and Stability of Glutamic Acid (Glu):
The glutamic acid component must maintain strict L-configuration, and its carboxyl groups require directional protection (e.g., OtBu) to preserve biological activity.
Leveraging deep expertise in peptide chemistry and industrial-scale manufacturing, Watson has successfully broken through the synthetic bottlenecks of these two key intermediates — AEEA series and glutamic acid derivatives — emerging as an “invisible champion” in global semaglutide API production.
I. AEEA Series Intermediates : From Molecular Design to Industrial Scale-Up
As the hydrophilic spacer in the side chain, the quality of AEEA units directly impacts drug solubility and metabolic behavior. Watson’s innovations have set new industry standards through :
1. Dual Activation Strategies for Flexible Supply
To address the diverse process requirements in semaglutide side chain synthesis, Watson offers two intermediate options :
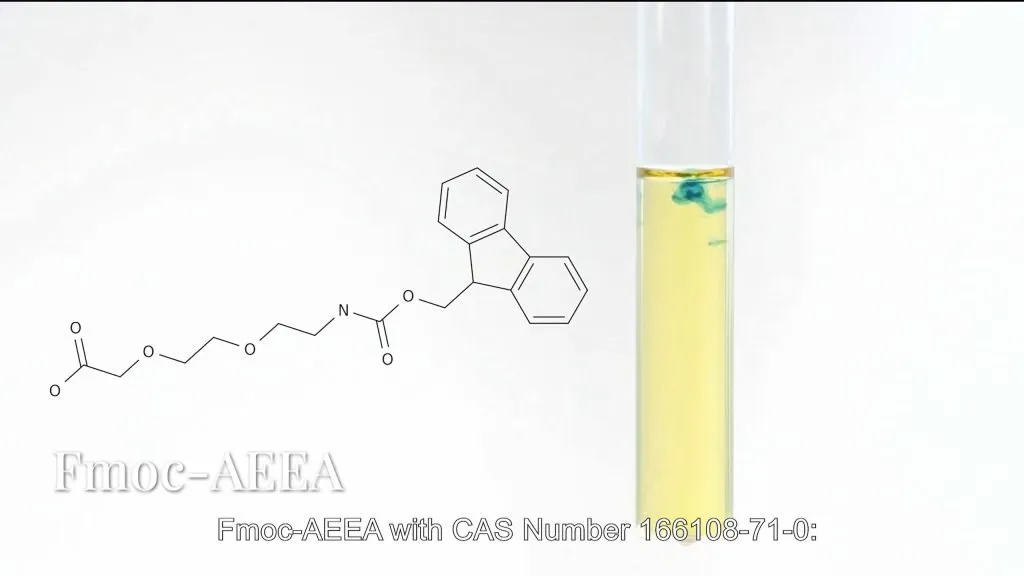
- Fmoc-AEEA (CAS : 166108-71-0):
Featuring Fmoc-protected amino and pre-activated carboxyl groups, this option allows direct use in solid-phase synthesis, eliminating 2-3 activation steps and shortening production cycles by over 30%.


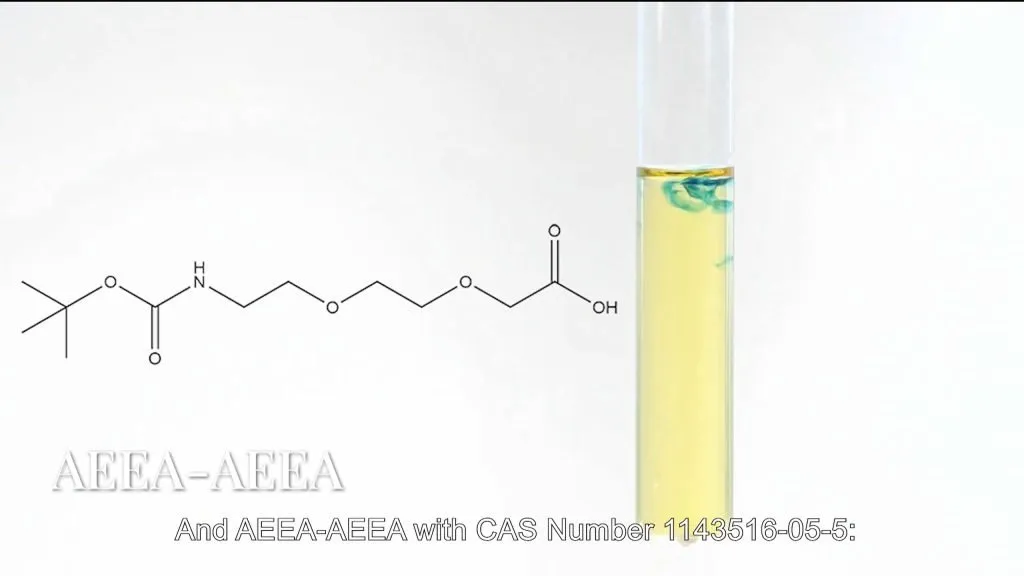
- AEEA-AEEA (CAS : 1143516-05-5):
Retains a non-activated hydroxyl end for custom activation approaches.


Both approaches leverage precision molecular engineering to ensure superior product consistency and performance.
2. Ultra-High Purity and Batch Consistency
Given the hygroscopic and oxidation-sensitive nature of AEEA intermediates, Watson employs “inert gas protection + low-temperature crystallization” purification processes to achieve purity >99.0% and limit single impurities to <0.3%. This stringent control enhances side chain synthesis yields by 15%-20% and minimizes the risk of batch failures due to intermediate variability.
3. GMP-Compliant Flexible Capacity for Global Demand
Watson’s GMP-certified facilities support seamless scalability from kilogram to ton levels, offering 30% faster lead times than the industry average and enabling customers to accelerate their entry into the GLP-1 drug market.
II. Glutamic Acid Derivatives : Breakthroughs in Stereocontrol and Protection Strategies
For precise coupling with AEEA-OSU modules, the glutamic acid derivative’s chemical and optical purity — as well as control of single impurities — are vital to preserving drug activity. Watson delivers solutions that directly address these industry pain points :
1. >99.0% Chemical Purity : The Foundation of Compliance and Cost Reduction
Semaglutide side chain synthesis demands >99.0% chemical purity, as residual trace impurities (e.g., oxidative byproducts) from lower-grade materials can amplify exponentially in downstream steps, jeopardizing entire batches. Watson employs gradient crystallization and supercritical chromatographic purification combined with a 200+ impurity database and end-to-end monitoring, ensuring purity exceeds 99.0% — enhancing coupling yields by 8%-12% and reducing purification costs.
2. 99.5% Optical Purity : Uncompromising Chiral Control
Watson’s Fmoc-Glu-OtBu (CAS : 84793-07-7) and Glu-OtBu (CAS : 45120-30-7) products achieve >99.5% optical purity through asymmetric synthesis and dynamic kinetic resolution. In-line process analytical technology (PAT) ensures batch-to-batch consistency in stereochemistry, completely eliminating risks of drug inactivation from isomer contamination.
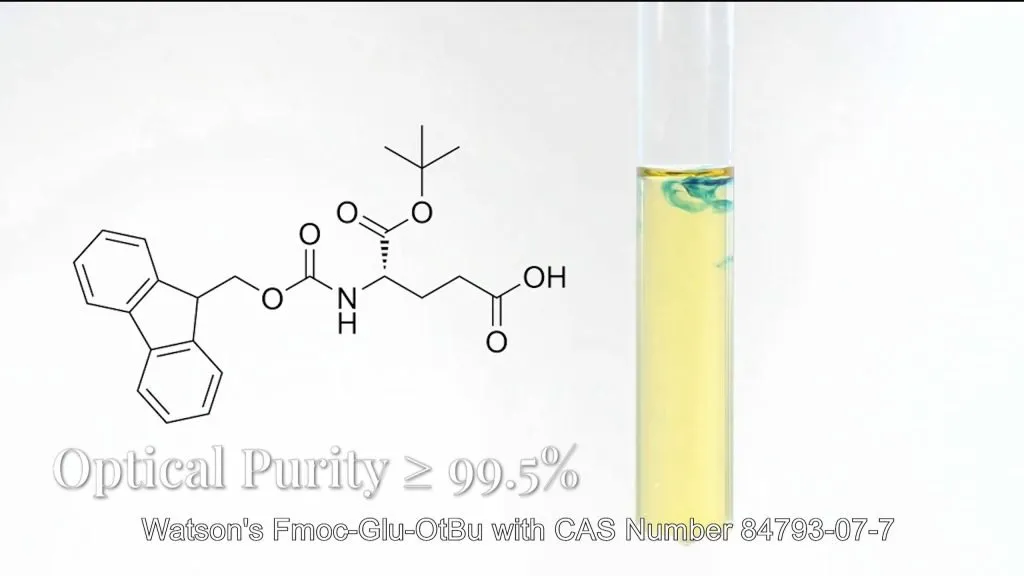


3. Tight Control of Single Impurities : Watson’s “Dual-Standard” Approach
While many suppliers focus solely on overall purity, neglecting the toxicological risks of specific impurities, Watson sets a new industry benchmark by limiting maximum single impurity to <0.3%, surpassing common specifications. For leading pharmaceutical companies requiring “zero tolerance” impurity levels, Watson offers customized <0.1% ultra-purity solutions without significant cost escalation.
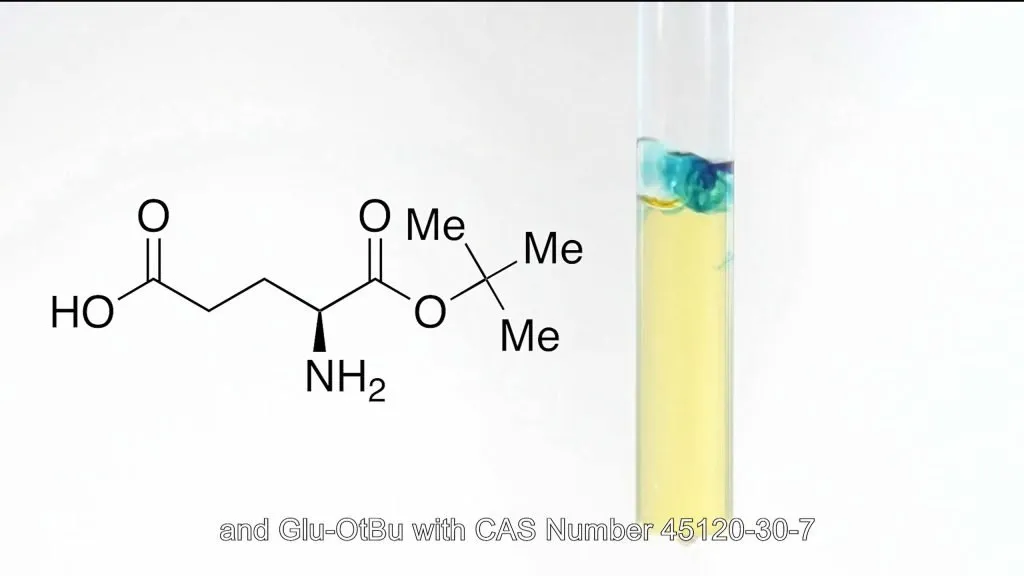


The global success of semaglutide underscores the immense potential of GLP-1 drugs in metabolic diseases. However, the quality and supply of side chain intermediates have become key competitive differentiators for pharmaceutical companies. Through technological innovation and industrial-scale breakthroughs in AEEA series and glutamic acid derivatives, Watson not only helps clients shorten R&D cycles by 6-12 months but also reduces API production costs by over 25%.
Looking ahead, Watson will continue to deepen its focus on peptide and PROTAC intermediates, driving the industry forward with its “molecular craftsmanship” spirit, and enabling innovative therapies to reach patients faster worldwide.
Related Products
Semaglutide intermediates on Watson