Identification
CAS Number
723-46-6
Name
ulfamethoxazole EP Impurity A2
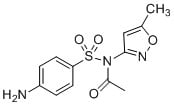
SMILES
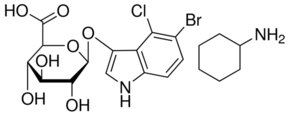
Cc1cc(no1)NS(=O)(=O)c2ccc(cc2)N
StdInChI
InChI=1S/C10H11N3O3S/c1-7-6-10(12-16-7)13-17(14,15)9-4-2-8(11)3-5-9/h2-6H,11H2,1H3,(H,12,13)
StdInChIKey
JLKIGFTWXXRPMT-UHFFFAOYSA-N
Molecular Formula
C10H11N3O3S
Molecular Weight
253.278
EINECS
211-963-3
MDL Number
MFCD00010546
Properties
Appearance
A white or almost white, crystalline powder.
Melting Point
169°C to 172°C
Infrared absorption spectrophotometry
The Infrared absorption Spectrum of sample is concordant with the spectrum of Sulfamethoxazole CRS.
Safety Data
Symbol
Signal Word
Warning
Hazard statements
H315-H319-H335Precautionary Statements
P305 + P351 + P338WGK Germany
3
Specifications and Other Information of Our
Identification Methods
HNMR, HPLC
Purity
99% min
Loss on Drying
NMT 0.5% w/w
Impurity A (Acetyl SMZ)
NMT 0.1 %
Impurity B (Sulfanilyl SMZ)
NMT 0.1 %
Impurity C (Isoxamine)
NMT 0.1 %
Impurity D (Sulfanilic acid)
NMT 0.1 %
Impurity E (Sulfanilamide)
NMT 0.1 %
Impurity F Isomeric (SMZ)
NMT 0.1 %
Any other Impurity
NMT 0.1 %
Total Impurities
NMT 0.3%
Shelf Life
5 years
Storage
Under room temperature away from light
Known Application
Sulfamethoxazole EP are mainly used to treat acute and chronic urinary tract infections, and can also be used to prevent meningitis and acute otitis media caused by influenza bacilli.
General View of Documents
Links
This product is developed by our R&D company Caming Pharmaceutical Ltd (https://www.caming.com/).
Quick Inquiry
Fill out our inquiry form and one of our experts will be in touch with you shortly.