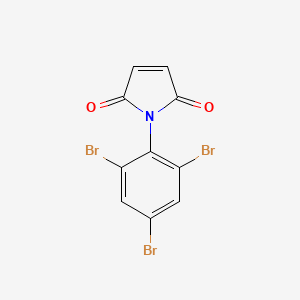
Identification
CAS Number
59789-51-4
Name
N-(2,4,6-Tribromophenyl)maleimide
Synonyms
1-(2,4,6-tribromophenyl)pyrrole-2,5-dione
SMILES
C1=CC(=O)N(C1=O)C2=C(C=C(C=C2Br)Br)Br
StdInChI
InChI=1S/C10H4Br3NO2/c11-5-3-6(12)10(7(13)4-5)14-8(15)1-2-9(14)16/h1-4H
StdInChIKey
ONEIBTGSNPDDSB-UHFFFAOYSA-N
Molecular Formula
C10H4Br3NO2
Molecular Weight
409.86
Properties
Appearance
Light yellow powder
Safety Data
RIDADR
NONH for all modes of transport
WGK Germany
3
Specifications and Other Information of Our N-(2,4,6-Tribromophenyl)maleimide CAS 59789-51-4
Identification Methods
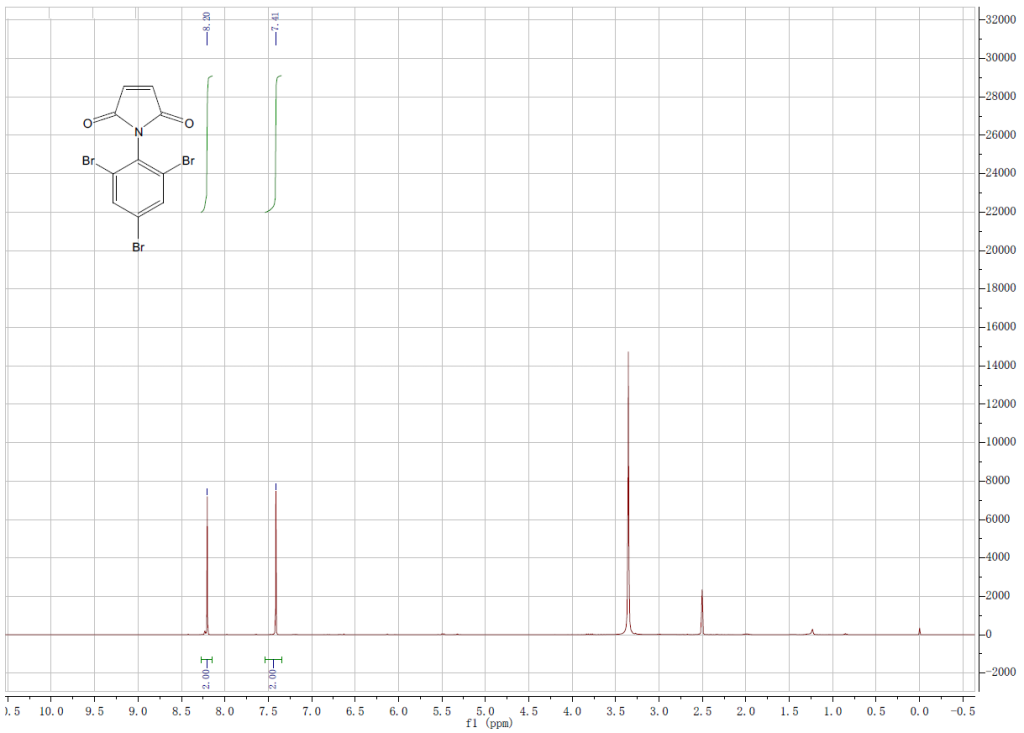
HNMR, HPLC
Purity
98% min
Assay
97%min
Shelf Life
2 years
Storage
Under room temperature away from light
Known Application
Can be used in aerospace composite materials (high temperature heat resistance); PCB electronic insulation materials (good flame retardancy); 3D printing photosensitive resins (high reactivity); high-performance coatings, adhesives (reactive structures)
- High-performance polymer additives or monomers
Used to synthesize special thermosetting resins or high-temperature resistant polymers, such as polyimide and maleimide copolymers ; its maleimide structure can undergo addition polymerization, free radical polymerization or Diels-Alder reaction with other monomers ; the aromatic tribromo substitution group gives the product flame retardancy and thermal stability, suitable for use in high-performance materials such as aviation, electronics, and insulation. - Flame retardant intermediates or synergists
The Br (bromine) element on the aromatic ring has excellent flame retardant effect ; this product can be used as a reactive flame retardant intermediate, embedded in the main chain or side chain of the polymer to improve the flame retardant efficiency and reduce migration ; it is often used for efficient flame retardant modification of polyester, polyamide, epoxy resin and other systems. - Electronic material intermediates
N-aryl maleimide compounds have excellent electronic properties and are widely used in : organic semiconductors, optoelectronic functional materials, photoresists and other functional polymer monomers. After tribromo substitution, it can be further derivatized, such as participating in Suzuki or Stille coupling to construct a π conjugated system. - Photosensitizer/crosslinker
Maleimide groups have good electron affinity and are often used in photo-initiated crosslinking reactions or UV curing systems. They can be rapidly photocrosslinked with thiols and alkenes to form a crosslinked network structure, which is used in photoresists, adhesives and other fields.
Product features
High thermal stability ; Excellent flame retardant properties ; Easy to modify ; High photo/thermal reactivity ; Good compatibility
General View of Documents


Links
This product is developed by our R&D company Warshel Chemical Ltd (https://www.warshel.com/).
Quick Inquiry
Fill out our inquiry form and one of our experts will be in touch with you shortly.