![Structure of Sulfonium, triphenyl-, salt with 2-sulfoethyl tricyclo[3.3.1.13,7]decane-1-carboxylate (11) <span class="caps">CAS</span> 1228823-28-6 Structure of Sulfonium, triphenyl-, salt with 2-sulfoethyl tricyclo[3.3.1.13,7]decane-1-carboxylate (11) CAS 1228823-28-6](https://www.watson-int.com/wp-content/uploads/2026/02/Structure-of-Sulfonium-triphenyl-salt-with-2-sulfoethyl-tricyclo3.3.1.137decane-1-carboxylate-11-CAS-1228823-28-6.png)
Identification
CAS Number
1228823-28-6
Name
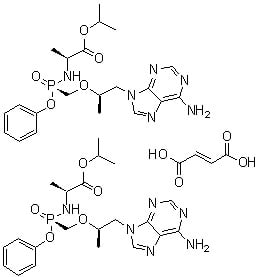
Sulfonium, triphenyl-, salt with 2-sulfoethyl tricyclo[3.3.1.13,7]decane-1-carboxylate (1:1)
Synonyms
Triphenylsulfonium 2-[(Adamantane-1-carbonyl)oxy]ethanesulfonate
triphenylsulfonium 2-(adamantane-1-carbonyloxy)ethanesulfonate
SMILES
C1C2CC3CC1CC(C2)(C3)C(=O)OCCS(=O)(=O)[O-].C1=CC=C(C=C1)[S+](C2=CC=CC=C2)C3=CC=CC=C3
StdInChI
InChI=1S/C18H15S.C13H20O5S/c1-4-10-16(11-5-1)19(17-12-6-2-7-13-17)18-14-8-3-9-15-18;14-12(18-1-2-19(15,16)17)13-6-9-3-10(7-13)5-11(4-9)8-13/h1-15H;9-11H,1-8H2,(H,15,16,17)/q+1;/p-1
StdInChIKey
WYYSEXOKTYXQKL-UHFFFAOYSA-M
Molecular Formula
C31H34O5S2
Molecular Weight
550.7
Properties
Appearance
White to off-white powder
Safety Data
RIDADR
NONH for all modes of transport
Specifications and Other Information of Our Sulfonium, triphenyl-, salt with 2-sulfoethyl tricyclo[3.3.1.13,7]decane-1-carboxylate (1:1) CAS 1228823-28-6
Identification Methods
HNMR, HPLC
Purity
99% min, 99.5% min
Total Heavy Metals Impurities
<100ppb,<50ppb
Shelf Life
2 years
Storage
Under room temperature away from light
Known Application
I. Core Function : Photoacid Generator (PAG)
This compound is a triphenylsulfonium salt-based photoacid generator (PAG). Under UV or deep-UV (DUV) irradiation :
It absorbs light and undergoes photolytic cleavage
Produces a strong acid (difluoromethanesulfonic acid)
The generated acid can catalyze chemically amplified reactions or initiate subsequent cationic polymerization. It is a key functional additive in photoresist and cationic photopolymer curing systems.
II. Main Application Areas
1.Semiconductor Photoresists (Chemically Amplified Photoresists, CARs)
Applicable to :
i-line, KrF, and ArF lithography systems
High-resolution chemically amplified photoresists
Main functions :
Acid generation upon exposure
Catalyzes deprotection reactions
Amplifies exposure effects → improves resolution, sensitivity, and critical dimension / line edge roughness (CD/LER) control
Advantages of the adamantyl structure :
Provides high thermal stability
Reduces acid diffusion → improves resolution and line edge roughness
Enhances optical performance and formulation stability of photoresists
- Cationic Photocuring Systems
Used in UV-induced cationic curing of epoxy resins and vinyl ether materials
UV irradiation → acid generation → initiates cationic ring-opening polymerization or crosslinking
Applications include :
UV-curable coatings and inks
Electronic encapsulation materials
Photo-patternable insulating layers (e.g., PSPI, PI)
Links
This product is developed by our R&D company WatsonChem Advanced Chemical Materials (https://www.watsonchem.com/).
Quick Inquiry
Fill out our inquiry form and one of our experts will be in touch with you shortly.

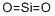
![Structure of 4-[2,6-dioxohexahydropyrimidin-4(S)-ylcarboxamido]-L-phenylalanyl-4-ureido-D-phenylalanyl-L-leucyl-N6-isopropyl-L-lysyl <span class="caps">CAS</span> <span class="caps">PNA-2731</span> Structure of 4-[2,6-dioxohexahydropyrimidin-4(S)-ylcarboxamido]-L-phenylalanyl-4-ureido-D-phenylalanyl-L-leucyl-N6-isopropyl-L-lysyl CAS PNA-2731](https://www.watson-int.com/wp-content/uploads/2019/09/Structure-of-4-26-dioxohexahydropyrimidin-4S-ylcarboxamido-L-phenylalanyl-4-ureido-D-phenylalanyl-L-leucyl-N6-isopropyl-L-lysyl-CAS-PNA-2731.png)