Identification
CAS Number
1392275-56-7
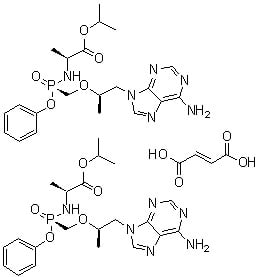
Name
Tenofovir Alafenamide Fumarate
Synonyms
(2E)-2-Butendisäure -isopropyl-N-[(S)-({[(2R)-1-(6-amino-9H-purin-9-yl)-2-propanyl]oxy}methyl)(phenoxy)phosphoryl]-L-alaninat (1:2) [German] [ACD/IUPAC Name]
1392275-56-7 [RN]
Acide (2E)-2-butènedioïque – N-[(S)-({[(2R)-1-(6-amino-9H-purin-9-yl)-2-propanyl]oxy}méthyl)(phénoxy)phosphoryl]-L-alaninate d’isopropyle (1:2) [French] [ACD/IUPAC Name]
GS-7340-03
Isopropyl N-[(S)-({[(2R)-1-(6-amino-9H-purin-9-yl)-2-propanyl]oxy}methyl)(phenoxy)phosphoryl]-L-alaninate (2E)-2-butenedioate (2:1) [ACD/IUPAC Name]
L-Alanine, N-[(S)-[(1S)-[(1R)-2-(6-amino-9H-purin-9-yl)-1-methylethoxy]methyl]phenoxyphosphinyl]-, 1-methylethyl ester, (2E)-2-butenedioate (2:1) [ACD/Index Name]
Tenofovir alafenamide fumarate [USAN]
bis{9-[(2R,5S,7E)-2,7,10-trimethyl-5,8-dioxo-5-phenoxy-3,9-dioxa-6-aza-5λ5-phosphaundecan-1-yl]-9H-purin-6-aminium} (2E)-but-2-enedioate ; bis{propan-2-yl N-[(S)-({[(2R)-1-(6-amino-9H-purin-9-yl)propan-2-yl]oxy}methyl)(phenoxy)phosphoryl]-L-alaninate} (2E)-but-2-enedioate
GS-7340 Hemifumarate
TAF
tenofovir alafenamide hemifumarate
UNII-FWF6Q91TZO
Vemlidy
SMILES
C[C@H](Cn1cnc2c1ncnc2N)OC[P@@](=O)(N[C@@H](C)C(=O)OC(C)C)Oc3ccccc3.C[C@H](Cn1cnc2c1ncnc2N)OC[P@@](=O)(N[C@@H](C)C(=O)OC(C)C)Oc3ccccc3.C(=C/C(=O)O)\C(=O)O
StdInChI
InChI=1S/2C21H29N6O5P.C4H4O4/c21-14(2)31-21(28)16(4)26-33(29,32-17-8-6-5-7-9-17)13-30-15(3)10-27-12-25-18-19(22)23-11-24-20(18)27;5-3(6)1-2-4(7)8/h25-9,11-12,14-16H,10,13H2,1-4H3,(H,26,29)(H2,22,23,24);1-2H,(H,5,6)(H,7,8)/b;;2-1+/t2*15-,16+,33+;/m11./s1
StdInChIKey
SVUJNSGGPUCLQZ-FQQAACOVSA-N
Molecular Formula
C46H62N12O14P2
Molecular Weight
1069.004
Properties
Appearance
White to almost White crystal or crystalline powder
Safety Data
WGK Germany
3
Specifications and Other Information of Our Tenofovir Alafenamide Fumarate CAS 1392275-56-7
Identification Methods
HPLC
Assay
97.0%~102.0%
Water
≤1.0%
Residue solvent
Acetonitrile : ≤0.041 %
Related substances
PMPA
≤1.0%
PMPAAnhydride
≤0.75%
PMPA Single phenyl PMPA
≤1.0%
Unknow single impurity
≤0.15%
Total Impurities
≤2.0%
Isomer
≤1.0%
Content of Fumaric acid
9.0%~13.0%
Shelf Life
2 years
Storage
Under room temperature away from light
Known Application
It is a new type of nucleotide reverse transcriptase inhibitor for the treatment of chronic hepatitis B virus infection and compensatory liver disease.

![Structure of 1,2,3,4-tetrahydro-benzo[h]quinolin-3-ol hydrochloride <span class="caps">CAS</span> 73579-26-7 Structure of 1,2,3,4-tetrahydro-benzo[h]quinolin-3-ol hydrochloride CAS 73579-26-7](https://www.watson-int.com/wp-content/uploads/2024/06/Structure-of-1234-tetrahydro-benzohquinolin-3-ol-hydrochloride-CAS-73579-26-7.png)