Identification
CAS Number
28053-08-9
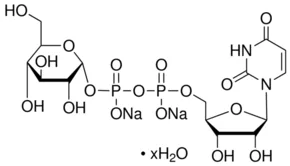
Name
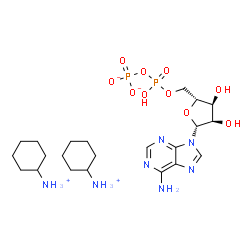
Uridine 5′-diphosphoglucose disodium salt
Synonyms
(3,4-Dihydroxytetrahydro-2-furanyl)methyl 3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl dihydrogen diphosphate [ACD/IUPAC Name]
(3,4-Dihydroxytetrahydro-2-furanyl)methyl-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yldihydrogendiphosphat [German] [ACD/IUPAC Name]
248-801-6 [EINECS]
28053-08-9 [RN]
Dihydrogénodiphosphate de (3,4-dihydroxytétrahydro-2-furanyl)méthyle et de 3,4,5-trihydroxy-6-(hydroxyméthyl)tétrahydro-2H-pyran-2-yle [French] [ACD/IUPAC Name]
SMILES
C1C(C(C(O1)COP(=O)(O)OP(=O)(O)OC2C(C(C(C(O2)CO)O)O)O)O)O
StdInChI
InChI=1S/C11H22O15P2/c12-1-5-8(15)9(16)10(17)11(24-5)25-28(20,21)26-27(18,19)23-3-6-7(14)4(13)2-22-6/h4-17H,1-3H2,(H,18,19)(H,20,21)
StdInChIKey
ZZAHNEVHQYWAEU-UHFFFAOYSA-N
Molecular Formula
C15H22N2Na2O17P2
Molecular Weight
610.26
EINECS
248-801-6
MDL Number
MFCD20441966
Properties
Appearance
White powder or off- white powder
Safety Data
Symbol
WGK Germany
3
MSDS Download
Specifications and Other Information of Our Uridine 5′-diphosphoglucose disodium salt CAS 28053-08-9
Identification Methods
HNMR, HPLC
Purity
98% min
Solution Clarity and Color
Clarify, colorless
Loss on Drying
≤10.0%
Shelf Life
2 years
Storage
Store at -20°C for long time, in container tightly sealed ; Protect from light.
Known Application
Intermediate in Carbohydrate Metabolism
UDP-Glc (Uridine Diphosphate Glucose) is an essential intermediate in carbohydrate metabolism, playing a crucial role particularly in glycogen and glycoprotein synthesis. As an activated form of glucose, it participates in glycosyl transfer reactions by transferring glucose units to other molecules.
Donor in Glycosylation Reactions
UDP-Glc serves as the primary donor in glycosyltransferase reactions and is frequently used in the synthesis of polysaccharides, glycolipids, and glycoproteins. In these reactions, UDP-Glc provides glucose groups, transferring them to acceptor molecules to facilitate complex carbohydrate structures.
Synthesis of Cell Wall and Extracellular Matrix
In plants, bacteria, and other organisms, UDP-Glc is involved in the synthesis of cell wall components such as cellulose and hemicellulose. This function is crucial for maintaining structural integrity, enabling cells to sustain and reinforce their structure.
Reagent in Metabolic Pathway Research
In biochemical and pharmacological research, UDP-Glc is widely used to study carbohydrate metabolism pathways, including glycogen metabolism and glycosylation reactions in cellular signaling pathways. It helps researchers investigate key biochemical processes and metabolic functions.
Precursor in Drug Metabolism
UDP-Glc also participates in the metabolism of certain drugs, especially in the liver, where it facilitates glycosylation reactions that increase the water solubility of drugs, making them easier to excrete from the body.
General View of Documents
Links
This product is developed by our R&D company Caming Pharmaceutical Limited (http://www.caming.com/).
Quick Inquiry
Fill out our inquiry form and one of our experts will be in touch with you shortly.