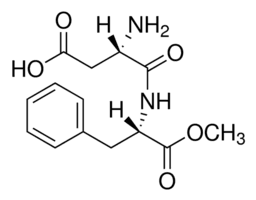
In the realm of modern biopharmaceutical innovation, antibody-drug conjugates (ADCs) represent a promising frontier — one that combines the specificity of monoclonal antibodies with the potency of cytotoxic agents. At the core of this technology lies a critical component : the linker, which bridges the antibody and the drug payload. Among the various types of linkers, polyethylene glycol (PEG) linkers have gained widespread prominence due to their unique physicochemical properties and ability to improve the pharmacokinetic profile of therapeutics.
As the industry continues to pursue more stable, effective, and targeted treatments, Watson International, the brand owner of ChemWhat®, has earned global recognition as a trusted manufacturer of high-purity PEG linkers — tailor-made to accelerate ADC development and commercialization.



The Role of PEG Linkers in ADCs
Polyethylene glycol (PEG) linkers serve several vital functions in the design of antibody-drug conjugates :
- Improved Solubility and Stability : PEG chains enhance water solubility of hydrophobic payloads and increase overall formulation stability.
- Controlled Drug Release : Functionalized PEG linkers enable precise control over payload release in the tumor microenvironment, minimizing systemic toxicity.
- Reduced Immunogenicity and Enhanced Circulation Time : PEGylation can mask immunogenic epitopes and extend circulation half-life, improving the therapeutic index.
- Versatility : PEG linkers are highly tunable in terms of length, molecular weight, and end-group chemistry, making them ideal for both cleavable and non-cleavable linker systems.
Due to these advantages, PEG linkers have become indispensable tools in the next generation of ADC platforms and are widely used in clinical candidates targeting various malignancies.



ChemWhat® Brand PEG Linkers : Setting a Global Standard
Watson’s ChemWhat® PEG linker series is specifically engineered for pharmaceutical-grade applications, with minimum purity levels exceeding 95%. Such high standards ensure consistent batch-to-batch performance, which is essential in tightly regulated environments like ADC clinical trials and commercial production.
Whether used in cleavable linkers (triggered by enzymatic or pH-sensitive mechanisms) or non-cleavable frameworks, ChemWhat® PEG linkers offer reproducibility, stability, and biocompatibility, aligning with stringent quality control systems adopted by global pharmaceutical firms.
Technical Strength and Customization Excellence
Watson’s strength lies not only in product purity, but also in its ability to provide custom-designed PEG linkers based on specific research needs. Backed by a dedicated R&D team with deep expertise in polymer chemistry, conjugation technologies, and controlled-release systems, Watson can deliver a wide array of PEG-based structures, including :
- Mono-, di-, and multi-arm PEGs
- Heterobifunctional PEG linkers (e.g., NHS-PEG-Maleimide)
- Acid-labile, enzymatically cleavable, or redox-sensitive PEG linkers
- Drug-payload compatible linkers with hydrophobic/hydrophilic tuning
These tailor-made services are crucial in helping ADC developers optimize the balance between stability and release kinetics, ultimately improving therapeutic outcomes.



Global Recognition by Leading Innovators
Watson has supplied PEG linkers to leading pharmaceutical companies and biotechs across the U.S., Germany, Japan, South Korea, and China, many of whom are engaged in pioneering ADC pipelines. The company has been selected repeatedly as a strategic supplier for its quality consistency, fast response time, and capability to meet both gram-scale R&D needs and multi-kilogram GMP-grade demands.
Long-term collaborations with global pharmaceutical companies highlight Watson’s commitment to innovation, reliability, and regulatory compliance. Whether it’s a multinational’s clinical stage ADC or a university’s proof-of-concept study, Watson’s products and services are trusted across the development spectrum.



Innovation, Quality, and Vision
With modern production facilities and cutting-edge analytical platforms, including HPLC, NMR, and LC-MS characterization, Watson ensures that every linker meets the strictest international standards. The company continues to invest in scalable manufacturing and green synthesis methods, aligning with both scientific progress and sustainable development goals.
As PEG linker chemistry continues to evolve alongside ADC technologies, Watson International remains at the forefront — not only as a supplier, but as a scientific partner — helping to bring targeted therapies from concept to clinic.
ChemWhat Reference Links
Available Polyethylene Glycol Products