Identification
CAS Number
1237826-25-3
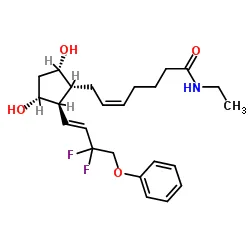
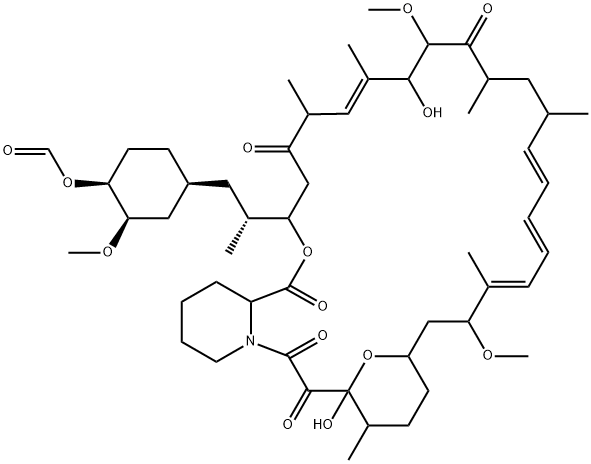
Name
Everolimus EP Impurity E
Synonyms
(1R,2R)-4-{(2R)-2-[(1R,9S,12S,15R,16E,18R,19R,21R,23S,24E,28E,30S,32S,35R)-1,18-Dihydroxy-19,30-dimethoxy-15,17,21,23,29,35-hexamethyl-2,3,10,14,20-pentaoxo-11,36-dioxa-4-azatricyclo[30.3.1.04,9]hex atriaconta-16,24,26,28-tetraen-12-yl]propyl}-2-methoxycyclohexyl formate [ACD/IUPAC Name]
(1R,2R)-4-{(2R)-2-[(1R,9S,12S,15R,16E,18R,19R,21R,23S,24E,28E,30S,32S,35R)-1,18-Dihydroxy-19,30-dimethoxy-15,17,21,23,29,35-hexamethyl-2,3,10,14,20-pentaoxo-11,36-dioxa-4-azatricyclo[30.3.1.04,9]hex atriaconta-16,24,26,28-tetraen-12-yl]propyl}-2-methoxycyclohexylformiat [German] [ACD/IUPAC Name]
23,27-Epoxy-3H-pyrido[2,1-c][1,4]oxaazacyclohentriacontine-1,5,11,28,29(4H,6H,31H)-pentone, 3-[(1R)-2-[(3R,4R)-4-(formyloxy)-3-methoxycyclohexyl]-1-methylethyl]-9,10,12,13,14,21,22,23,24,25,26,27,32,3 3,34,34a-hexadecahydro-9,27-dihydroxy-10,21-dimethoxy-6,8,12,14,20,26-hexamethyl-, (3S,6R,7E,9R,10R,12R,14S,15E,19E,21S,23S,26R,27R,34aS)- [ACD/Index Name]
Formiate de (1R,2R)-4-{(2R)-2-[(1R,9S,12S,15R,16E,18R,19R,21R,23S,24E,28E,30S,32S,35R)-1,18-dihydroxy-19,30-diméthoxy-15,17,21,23,29,35-hexaméthyl-2,3,10,14,20-pentaoxo-11,36-dioxa-4-azatricyclo[30.3. ;1.04,9]hexatriaconta-16,24,26,28-tétraén-12-yl]propyl}-2-méthoxycyclohexyle [French] [ACD/IUPAC Name]
1237826-25-3 [RN]
SMILES
CC1=CC=CC=C[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)C(C)=C[C@@H](C)C(=O)C[C@H](OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@]2(O)O[C@H](C[C@@H]1OC)CC[C@H]2C)[C@H](C)CC1C[C@@H](OC)[C@@H](CC1)OC=O |c:1,t:5,20|
StdInChI
InChI=1S/C52H79NO14/c1-31-16-12-11-13-17-32(2)43(62-8)28-39-21-19-37(7)52(61,67-39)49(58)50(59)53-23-15-14-18-40(53)51(60)66-44(34(4)26-38-20-22-42(65-30-54)45(27-38)63-9)29-41(55)33(3)25-36(6)47(57)48(64-10)46(56)35(5)24-31/h11-13,16-17,25,30-31,33-35,37-40,42-45,47-48,57,61H,14-15,18-24,26-29H2,1-10H3/b13-11?,16-12+,32-17+,36-25+/t31-,33-,34-,35-,37-,38?,39+,40+,42-,43+,44+,45-,47-,48+,52-/m1/s1
StdInChIKey
IUPNCBWADBCXHV-XONPCIIHSA-N
Molecular Formula
C52H79NO14
Molecular Weight
942.182
Properties
Appearance
White Solid
Safety Data
WGK Germany
3
Specifications and Other Information of Our Everolimus EP Impurity E CAS 1237826-25-3
Identification Methods
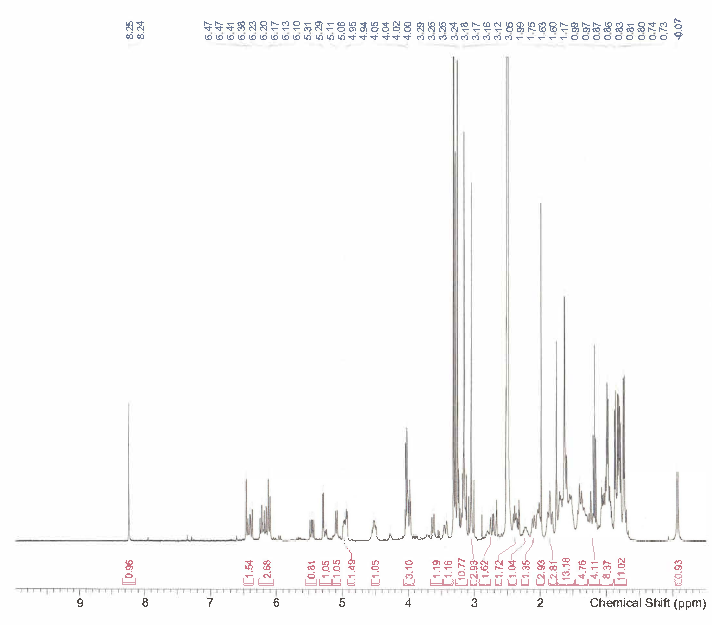
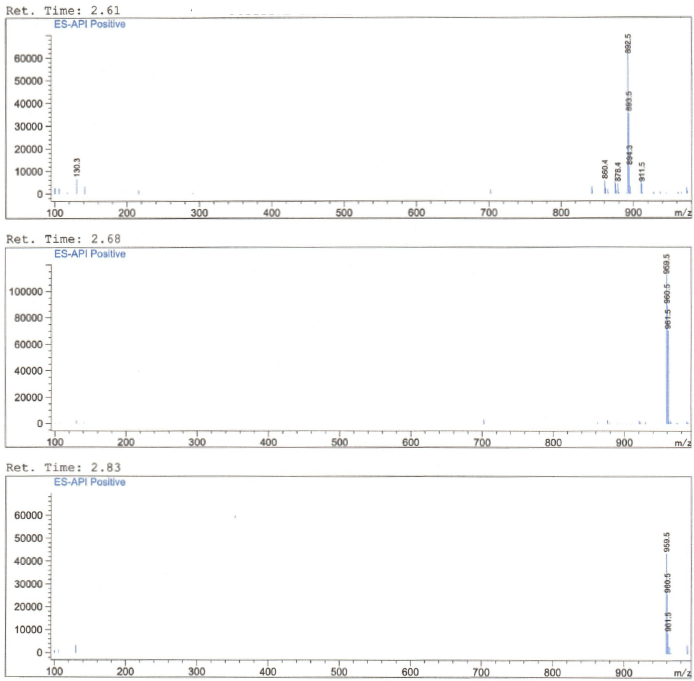
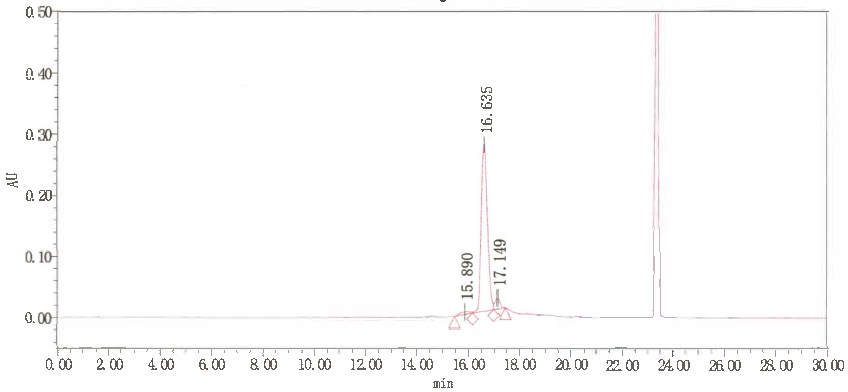
HNMR, HPLC
Purity
≥95%
Assay
≥90%
Shelf Life
3 years
Storage
Store at 2-8℃, sealed and away from light.
Known Application
Used as the Impurity E of Everolimus.
General View of Documents
Links
This product is developed by our R&D company Caming Pharmaceutical Ltd (https://www.caming.com/).
Quick Inquiry
Fill out our inquiry form and one of our experts will be in touch with you shortly.